The Supplement That's Everywhere Right Now
If you follow supplement discourse at all, you've encountered apigenin in the last year. It's the third leg of the Huberman sleep cocktail: 50 mg nightly alongside magnesium threonate and theanine. It shows up in longevity stacks positioned as a way to preserve NAD⁺ levels as you age. And more recently, it's appearing in senolytic formulas marketed for cellular aging, usually paired with fisetin or quercetin.
Three claims. Three consumer categories. One compound pulled from chamomile, parsley, and celery.¹
The biology behind each claim is real, and it's been studied for over thirty years.²,³ Apigenin is one of the most pharmacologically interesting flavonoids in the literature. But when you line up the claims people are buying it for against the evidence that exists in humans, the two lists don't match.
Three Mechanisms, Three Reasons It Sounds Right
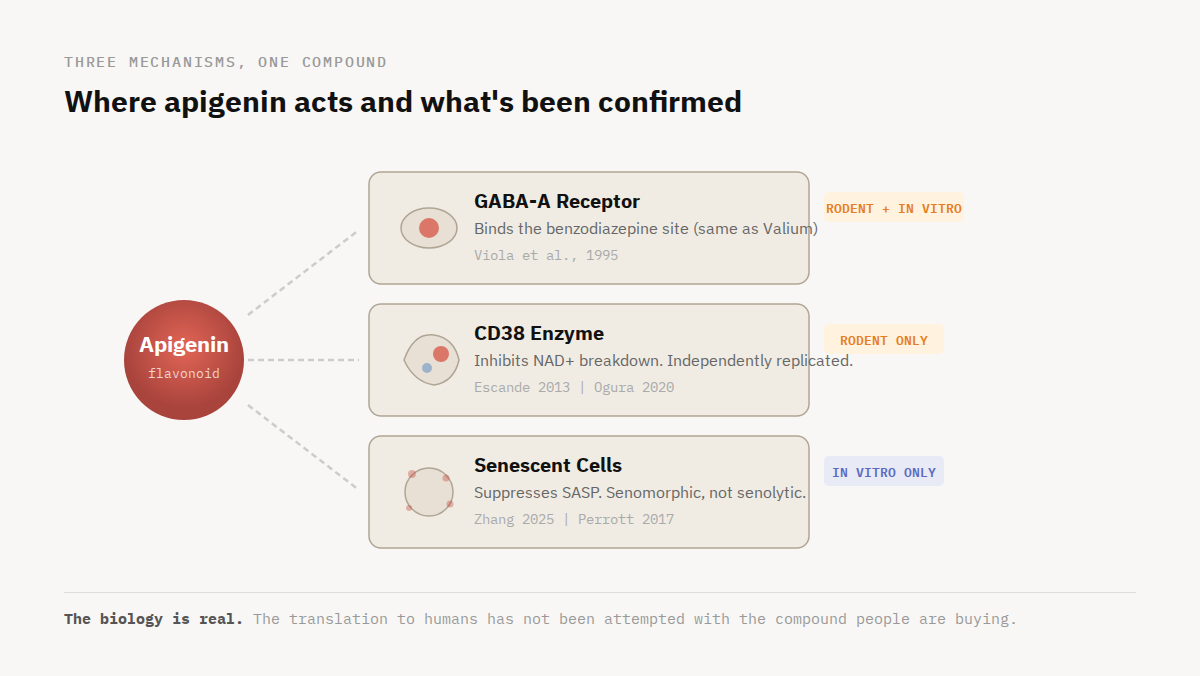
The biology behind each claim is not invented. That's part of what makes apigenin interesting. Many trending supplements lean on one mechanism with one or two papers behind it. Apigenin has three distinct pathways that apigenin users and sellers like to lean on, each with a plausible rationale.
The calming pathway. Your brain's primary braking system runs on a neurotransmitter called GABA. When GABA binds its receptor, it slows neuronal firing. That's how your brain quiets down. Benzodiazepines like Valium work by amplifying this braking signal: they bind a specific site on the GABA receptor and make the brake more sensitive.
In 1995, Viola and colleagues in Buenos Aires showed that apigenin, isolated from chamomile flowers, binds the same site.² Not vaguely, not through some indirect antioxidant effect. It's a competitive ligand at the benzodiazepine binding pocket, confirmed through displacement assays and blocked by the same antagonist that reverses Valium.² At low concentrations, the result in rodent models is calming without sedation. At higher concentrations, sedation sets in. That profile is consistent with a partial agonist that doesn't fully activate the receptor the way a pharmaceutical benzodiazepine does.
If you're a supplement brand and you have a compound that binds the same receptor as Valium but doesn't carry the dependence risk, the sleep pitch writes itself.
The NAD⁺ pathway. NAD⁺ is a molecule every cell in your body uses for energy production, DNA repair, and cellular maintenance. Its levels decline measurably with age, across human skin, brain, and blood. The primary driver of that decline is an enzyme called CD38. It breaks down NAD⁺, and its expression has been shown to rise in various tissues with age. More CD38 usually means less NAD⁺, which means less fuel for the repair processes that are supposed to run overnight.
Most longevity supplements try to solve this by adding more NAD⁺ precursors: NMN, NR, niacin. In 2013, Escande and colleagues at the Mayo Clinic showed that apigenin takes the other side of the equation.³ It directly inhibits CD38. Instead of pouring more water into the tub (via precursor administration), it partially closes the drain. That framing, block the enzyme that's destroying your NAD⁺ instead of just making more, resonated immediately with the longevity community. And the finding didn't sit in isolation. In 2020, Ogura's group in Japan reproduced the core result in a different species, a different organ, and a different disease model.⁸ Two labs, two countries, same conclusion. That kind of independent replication is what makes the mechanism credible.
The senescence pathway. As cells accumulate damage over a lifetime, some stop dividing but refuse to die. These senescent cells build up in tissues and leak a cocktail of inflammatory signals, collectively called the SASP, that damage surrounding healthy cells.¹⁰ The longevity field has spent the last decade looking for compounds that address this. Senolytics are designed to clear senescent cells. Senomorphics take a different approach: they quiet the inflammatory output without clearing the cells.
In 2025, Zhang and colleagues screened 66 natural compounds and identified apigenin as a senomorphic.¹⁰ It suppresses the SASP. It does not clear the cells. That distinction matters, because most of the products it appears in are marketed as senolytics. The biology says one thing. The label says another.
Each of these pathways is supported by real pharmacology from real research groups. None of them are marketing fiction. The question is simpler and harder: what happened when someone actually tested this in people if it was tested at all?
What Happened When Someone Checked
The calming pathway: tested in humans, but not the way you'd think. The most important thing to know about the human evidence for apigenin is that none of it tested apigenin. Not by itself anyway. Every controlled trial used chamomile extract, a full botanical matrix containing dozens of active compounds, standardized to contain a small amount of apigenin (up to 1.2% apigenin by weight).
Amsterdam and colleagues ran the first trial in 2009.⁵ Fifty-seven adults with generalized anxiety disorder received chamomile extract or placebo for eight weeks. The dose was flexible, adjusted per patient, delivering roughly 3 to 13 mg of apigenin per day. Anxiety scores dropped significantly compared to placebo. Small trial, modest effect, but it was the first evidence that apigenin might be calming in humans.⁵
Mao and colleagues followed up in 2016 with a larger and longer study.⁶ One hundred seventy-nine generalized anxiety patients took 1,500 mg of chamomile extract daily, delivering about 18 mg of apigenin, for 12 weeks. About half responded. Those 93 responders were then randomized: continue chamomile or switch to placebo for another 26 weeks. Here is the part that rarely gets mentioned. The study was designed to test whether chamomile prevented anxiety relapse. It did not. The primary endpoint missed. What the study did find, as a secondary outcome, was that patients who stayed on chamomile maintained lower anxiety symptoms throughout follow-up. That's a meaningful finding. But a secondary result in a trial that missed its primary endpoint is not the same thing as a definitive positive, and it's almost never presented that way.
For sleep specifically, Zick and colleagues tested chamomile extract in 34 adults with chronic insomnia for 28 days.⁴ No significant difference on any sleep measure was found. Sleep latency and nighttime awakenings showed moderate effect sizes favoring chamomile, but total sleep time actually favored placebo.⁴ This is the only controlled trial that directly measured sleep, and it was null.
Godos and colleagues published an observational study in 2020 surveying dietary polyphenol intake across nearly 2,000 Italian adults.¹¹ Higher dietary apigenin intake correlated with better sleep quality, but the study measured diet, not supplementation, and apigenin was one of several polyphenols that showed the association.¹¹ This is a dietary correlation, not evidence that an apigenin capsule improves sleep.
In 2026, Wong and colleagues tested part of the combination made popular by Andrew Huberman, magnesium (thought the type was not specified) and apigenin together, in three mouse models of disrupted sleep.⁷ Apigenin alone did not significantly improve sleep in any model. Magnesium alone did not either. The combination produced a synergistic effect, increasing sleep duration 32 to 44 percent across all three models. Even in the most favorable preclinical study for sleep, apigenin didn't carry the sleep effect by itself.
The NAD⁺ pathway: replicated in animals, absent in humans. Escande's 2013 finding at the Mayo Clinic remains the foundation.³ Obese mice received apigenin by intraperitoneal injection at 100 mg/kg daily for one week. Liver NAD⁺ rose. Metabolic markers improved. The mechanism was specific: when the experiment was repeated in cells lacking CD38, apigenin had no effect on NAD⁺, confirming it works through CD38 inhibition and not some other route.
But the same dose tells a different story in a different context. In 2012, Singh and colleagues administered apigenin to mice at 25, 50, 100, and 200 mg/kg by the same route.¹³ At 100 mg/kg, the dose Escande used, they found elevated liver enzymes, increased oxidative stress, and damaged liver tissue. Below 50 mg/kg, no toxicity was observed.¹³ Same compound, same species, same injection route, opposite findings in the liver. That doesn't invalidate the CD38 mechanism, but it puts a hard ceiling on the dose range anyone should be extrapolating from. Even if this dose was oral, it would be orders of magnitude higher than what someone would (or should be) consuming as a supplement.
Now, interestingly, Ogura's group in Japan reproduced the core CD38 finding in 2020 in a different species (rats), a different organ (kidney), a different disease (diabetic kidney disease), at one-fifth the dose (20 mg/kg), given orally instead of by injection.⁸ That lower dose and oral route matter. It suggests the mechanism may not require the heroic exposure that produced both Escande's metabolic improvements and Singh's liver damage.
The dose question does not get easier from there. Even Ogura's lower oral dose translates to over 200 mg in a human using standard FDA allometric scaling.8 A typical supplement capsule contains 50 mg. How much of that reaches systemic circulation is genuinely unknown, because no pharmacokinetic study has ever been published for isolated oral apigenin in humans. No one has measured plasma levels after a dose. No one has measured intracellular NAD⁺ levels after oral apigenin at any dose, for any duration. The entire dosing rationale for the NAD⁺ claim is built on animal estimates using doses higher than just about anyone is using as a supplement.
The senescence pathway: new, well-designed, preclinical only. Zhang's 2025 screen is the primary data here.¹⁰ Out of 66 natural compounds tested for both senolytic and senomorphic activity, apigenin emerged as a senomorphic agent. It suppresses the inflammatory output of senescent cells. It does not clear them. Earlier work from the Campisi laboratory in 2017 had shown similar SASP suppression across multiple human fibroblast strains and senescence triggers, without inducing apoptosis, and demonstrated that quieting those senescent cells reduced the aggressive behavior of nearby breast cancer cells.¹² Both groups point in the same direction: apigenin quiets senescent cells without killing them. No human has ever been tested.
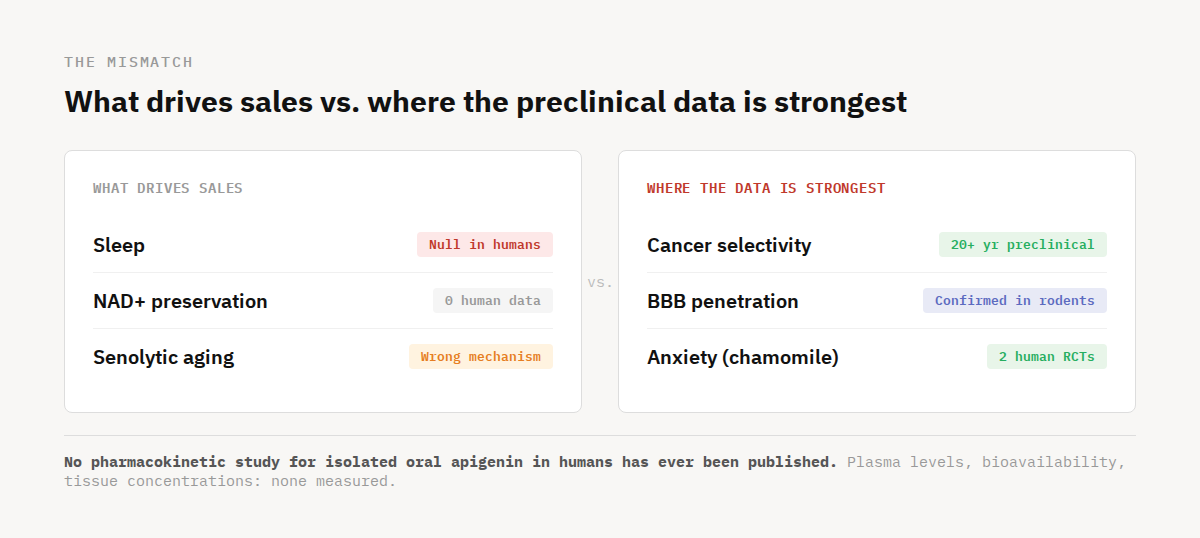
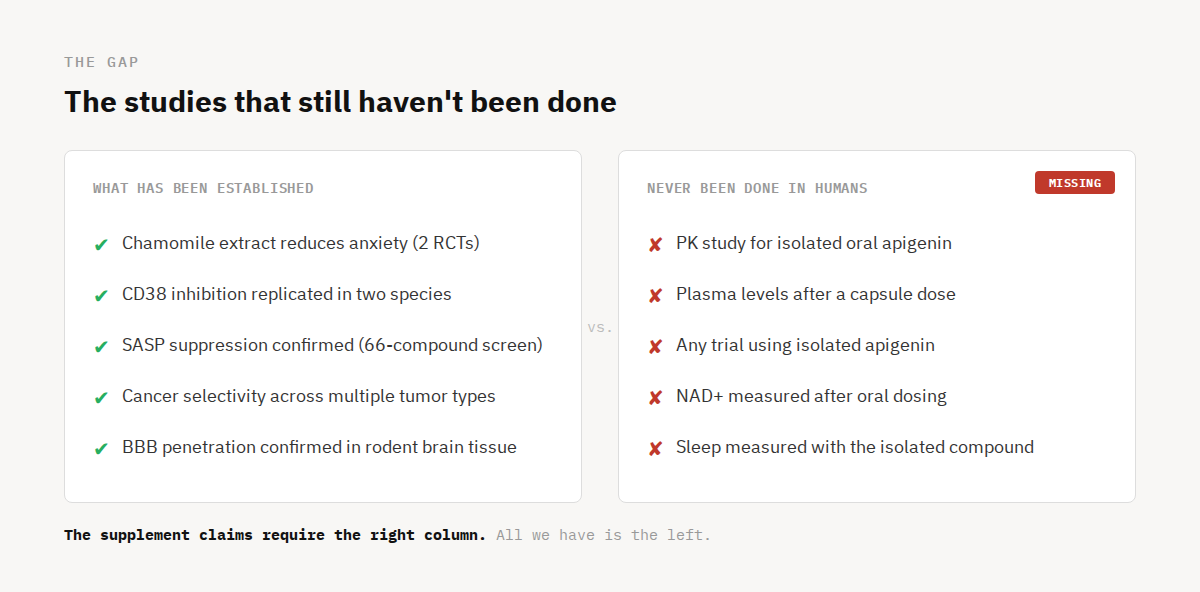
One pattern cuts across all three domains. Every controlled human trial that showed a positive result used chamomile extract, not isolated apigenin. The supplement market sells isolated apigenin at 50 mg per capsule, citing trials that tested a different compound at a different dose. Whether isolated apigenin by itself produces the same effects in a human body is a question that has never been asked in a clinical trial.

What the Hype Missed
There are two things the current apigenin conversation gets wrong, and they point in opposite directions.
The first is an absence. No one has ever published a pharmacokinetic study for isolated oral apigenin in humans. No one has ever published a pharmacokinetic study for isolated oral apigenin in humans. The closest data comes from a 2022 study that measured apigenin metabolites after chamomile tea and parsley consumption, not supplement capsules, and found that free apigenin barely reached circulation, with urinary metabolites accounting for just 0.5% of intake.¹⁴ What happens when someone swallows 50 mg of isolated apigenin in a capsule has never been measured.
The second is a set of findings the market walked past. While the consumer conversation fixates on sleep and NAD⁺, the preclinical domains where apigenin's data is most distinctive barely appear on any label.
The largest body of preclinical research on apigenin has nothing to do with sleep or NAD⁺. It's cancer. Two decades of consistent findings across renal, colorectal, breast, and prostate cancer cell lines showing cell cycle arrest and selective killing of cancer cells while sparing normal tissue.¹ That selectivity, damaging cancer cells without harming healthy ones, is what drew researchers to the molecule in the first place. Two Phase II clinical trials were initiated to test this in humans. Both were suspended before generating data.¹ What this means for apigenin and its utility in humans in the fight against cancer is uncertain. Again, the preclinical evidence is promising (and consistent). The human evidence is what’s missing.
The brain data is similarly underappreciated. Most polyphenols don't cross the blood-brain barrier in meaningful quantities. Apigenin might, at least in rodents. In 2013, Zhao and colleagues gave it orally to transgenic Alzheimer's mice for three months and found it rescued spatial memory and reduced amyloid plaque burden in the brain.¹⁶ A separate study measured apigenin directly in rat brain tissue after injection, confirming it can cross the blood-brain barrier, though whether oral dosing achieves the same brain concentrations is unconfirmed.¹⁷ That combination of brain effects after oral dosing and confirmed BBB penetration after injection is more than most polyphenols can claim, even if the gap between the two hasn't been closed.
None of this is proven in humans. All of it is preclinical. But the cancer selectivity data alone span two decades of independent replication across multiple tumor types; the neuroprotection findings come from several different disease models. These are not fringe findings. They're just not the findings driving sales.
What the Evidence Supports and What It Doesn't
Apigenin is not a bad molecule with good marketing. It's a genuinely interesting molecule with misaligned marketing.
What's established: chamomile extract delivering 13 to 18 mg of apigenin per day reduces anxiety in adults with generalized anxiety disorder across two controlled trials, 236 patients, and treatment periods up to 38 weeks.⁵ ⁶ That is the only replicated human finding. It measured anxiety, not sleep. It tested chamomile, not isolated apigenin.
What's preclinically credible: CD38 inhibition independently replicated across two labs, two species, and two disease models.³ ⁸ Senomorphic activity identified through a well-designed 66-compound screen and consistent with earlier Campisi laboratory findings.¹⁰ ¹² Cancer selectivity spanning two decades across multiple tumor types.¹ Cognitive rescue in Alzheimer's mice after oral dosing.¹⁶ These are real findings. They are not marketing constructions.
What's missing: a human pharmacokinetic study for isolated apigenin. A controlled trial using the isolated compound. A single measurement of NAD⁺ in a human after oral dosing. These are not exotic asks. They are the most basic steps in translating any compound from animal models to clinical use, and none of them have been taken.
Apigenin has earned serious scientific attention. It has not yet earned the claims awarded to it by the internet.
References:
1. Salehi B, Venditti A, Sharifi-Rad M, et al. The therapeutic potential of apigenin. Int J Mol Sci. 2019;20(6):1305.
2. Viola H, Wasowski C, Levi de Stein M, et al. Apigenin, a component of Matricaria recutita flowers, is a central benzodiazepine receptors-ligand with anxiolytic effects. Planta Med. 1995;61(3):213-6.
3. Escande C, Nin V, Price NL, et al. Flavonoid apigenin is an inhibitor of the NAD⁺ase CD38: implications for cellular NAD⁺ metabolism, protein acetylation, and treatment of metabolic syndrome. Diabetes. 2013;62(4):1084-93.
4. Zick SM, Wright BD, Sen A, Arnedt JT. Preliminary examination of the efficacy and safety of a standardized chamomile extract for chronic primary insomnia. BMC Complement Altern Med. 2011;11:78.
5. Amsterdam JD, Li Y, Soeller I, et al. A randomized, double-blind, placebo-controlled trial of oral Matricaria recutita (chamomile) extract therapy for generalized anxiety disorder. J Clin Psychopharmacol. 2009;29(4):378-82.
6. Mao JJ, Xie SX, Keefe JR, et al. Long-term chamomile (Matricaria chamomilla L.) treatment for generalized anxiety disorder: a randomized clinical trial. Phytomedicine. 2016;23(14):1735-42.
7. Wong WY, Dow Chan B, Zhou H, Leung TC, Tai WCS. Synergistic sleep-promoting effects of magnesium and apigenin in normal and insomnia mouse models. Food Funct. 2026;17:2225-39.
8. Ogura Y, Kitada M, Xu J, Monno I, Koya D. CD38 inhibition by apigenin ameliorates mitochondrial oxidative stress through restoration of the intracellular NAD⁺/NADH ratio and Sirt3 activity in renal tubular cells in diabetic rats. Aging. 2020;12:11325-36.
9. Gradolatto A, Canivenc-Lavier MC, Basly JP, et al. Pharmacokinetics and metabolism of apigenin in female and male rats after a single oral administration. Drug Metab Dispos. 2005;33(2):203-8.
10. Zhang H, Xu Q, Jiang Z, et al. Targeting senescence with apigenin improves chemotherapeutic efficacy and ameliorates age-related conditions in mice. Adv Sci. 2025;12(20):e2412950.
11. Godos J, Ferri R, Castellano S, et al. Specific dietary (poly)phenols are associated with sleep quality in a cohort of Italian adults. Nutrients. 2020;12(5):1226.
12. Perrott KM, Wiley CD, Desprez PY, Campisi J. Apigenin suppresses the senescence-associated secretory phenotype and paracrine effects on breast cancer cells. Food Funct. 2017;8(2):733-43.
13. Singh P, Mishra SK, Noel S, Sharma S, Rath SK. Acute exposure of apigenin induces hepatotoxicity in Swiss mice. PLOS One. 2012;7(2):e31964.
14. Borges G, Fong RY, Ensunsa JL, Kimball J, Medici V, Ottaviani JI, Crozier A. Absorption, distribution, metabolism and excretion of apigenin and its glycosides in healthy male adults. Free Radic Biol Med. 2022;185:90-96.
15. Lauritzen KH, Yang K, Frisk M, Louwe MC, Olsen MB, Ziegler M, Louch WE, Halvorsen B, Aukrust P, Yndestad A, Sandanger Ø. Apigenin inhibits NLRP3 inflammasome activation in monocytes and macrophages independently of CD38. Front Immunol. 2024;15:1497984.
16. Zhao L, Wang JL, Liu R, et al. Neuroprotective, anti-amyloidogenic and neurotrophic effects of apigenin in an Alzheimer's disease mouse model. Molecules. 2013;18(8):9949-65.
17.Popović M, et al. Distribution of apigenin in rat tissues following oral and intraperitoneal administration. Acta Pharmaceutica. 2014;64(4):489-500.